

Function and mechanism of PIWI/piRNA in cancer
Received date: 2021-06-16
Online published: 2021-10-22
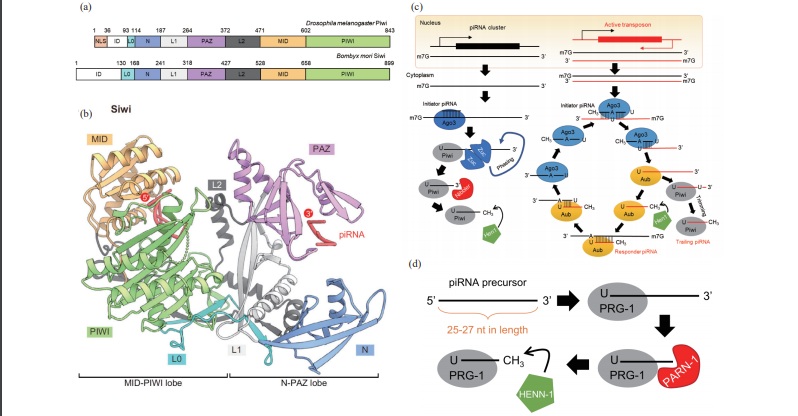
Key words: ; PIWI, piRNA, cancer
SHI Shuo, WANG Chen, LIN Haifan . Function and mechanism of PIWI/piRNA in cancer[J]. Chinese Journal of Nature, 2021 , 43(5) : 313 -322 . DOI: 10.3969/j.issn.0253-9608.2021.05.001
[1] SONG J J, SMITH S K, HANNON G J, et al. Crystal structure of Argonaute and its implications for RISC slicer activity [J]. Science, 2004, 305(5689): 1434-1437.
[2] MATSUMOTO N, NISHIMASU H, SAKAKIBARA K, et al. Crystal structure of silkworm PIWI-clade Argonaute Siwi bound to piRNA [J]. Cell, 2016, 167(2): 484-497.
[3] WANG C, LIN H. Roles of piRNAs in transposon and pseudogene regulation of germline mRNAs and lncRNAs [J]. Genome Biol, 2021, 22(1): 27.
[4] GUNAWARDANE L S, KUNIAKI S, KAZUMICHI M N, et al. A slicer-mediated mechanism for repeat-associated siRNA 5' end formation in Drosophila [J]. Science, 2007, 315(5818): 1587-1590.
[5] OKAMURA K, ISHIZUKA A, SIOMI H, et al. Distinct roles for Argonaute proteins in small RNA-directed RNA cleavage pathways [J]. Genes Dev, 2004, 18(14): 1655-1666.
[6] VAGIN V V, SIGOVA A, LI C, et al. A distinct small RNA pathway silences selfish genetic elements in the germline [J]. Science, 2006, 313(5785): 320-324.
[7] KIM V N, HAN J, SIOMI M C. Biogenesis of small RNAs in animals [J]. Nat Rev Mol Cell Biol, 2009, 10(2): 126-139.
[8] TEIXEIRA F K, OKUNIEWSKA M, MALONE C D, et al. piRNAmediated regulation of transposon alternative splicing in the soma and germ line [J]. Nature, 2017, 552(7684): 268-272.
[9] LEWIS S H, QUARLES K A, YANG Y, et al. Pan-arthropod analysis reveals somatic piRNAs as an ancestral defence against transposable elements [J]. Nat Ecol Evol, 2018, 2(1): 174-181.
[10] ROSS R J, WEINER M M, LIN H. PIWI proteins and PIWIinteracting RNAs in the soma [J]. Nature, 2014, 505(7483): 353-359.
[11] ARAVIN A, GAIDATZIS D, PFEFFER S, et al. A novel class of small RNAs bind to MILI protein in mouse testes [J]. Nature, 2006, 442(7099): 203-207.
[12] GIRARD A, SACHIDANANDAM R, HANNON G J, et al. A germline-specific class of small RNAs binds mammalian Piwi proteins [J]. Nature, 2006, 442(7099): 199-202.
[13] GRIVNA S T, BEYRET E, WANG Z, et al. A novel class of small RNAs in mouse spermatogenic cells [J]. Genes Dev, 2006 20(13): 1709-1714.
[14] LAU N C, SETO A G, KIM J, et al. Characterization of the piRNA complex from rat testes [J]. Science, 2006, 313(5785): 363-367.
[15] GU W, LEE H C, CHAVES D, et al. CapSeq and CIP-TAP identify Pol II start sites and reveal capped small RNAs as C. elegans piRNA precursors [J]. Cell, 2012, 151(7): 1488-1500.
[16] RUBY J G, JAN C, PLAYER C, et al. Large-scale sequencing reveals 21U-RNAs and additional microRNAs and endogenous siRNAs in C. elegans [J]. Cell, 2006, 127(6): 1193-1207.
[17] BRENNECKE J, ARAVIN A A, STARK A, et al. Discrete small RNA-generating loci as master regulators of transposon activity in Drosophila [J]. Cell, 2007, 128(6): 1089-1103.
[18] SAXE J P, CHEN M, ZHAO H, et al. Tdrkh is essential for spermatogenesis and participates in primary piRNA biogenesis in the germline [J]. EMBO J, 2013, 32(13): 1869-1885.
[19] KAWAOKA S, IZUMI N, KATSUMA S, et al. 3' end formation of PIWI-interacting RNAs in vitro [J]. Mol Cell, 2011, 43(6):1015- 1022.
[20] TANG W, TU S, LEE H C, et al. The RNase PARN-1 trims piRNA 3'ends to promote transcriptome surveillance in C. elegans [J]. Cell, 2016, 164(5): 974-984.
[21] IZUMI N, SHOJI K, SAKAGUCHI Y, et al. Identification and functional analysis of the pre-piRNA 3' trimmer in silkworms [J]. Cell, 2016, 164(5): 962-973.
[22] HOMOLKA D, PANDEY R R, GORIAUX C, et al. PIWI slicing and RNA elements in precursors instruct directional primary piRNA biogenesis [J]. Cell Rep, 2015, 12(3): 418-428.
[23] MOHN F, HANDLER D, BRENNECKE J. Noncoding RNA. piRNA-guided slicing specifies transcripts for Zucchini-dependent, phased piRNA biogenesis [J]. Science, 2015, 348(6236): 812-817.
[24] HAN B W, WANG W, LI C, et al. Noncoding RNA. piRNA-guided transposon cleavage initiates Zucchini-dependent, phased piRNA production [J]. Science, 2015, 348(6236): 817-821.
[25] HORWICH M D, LI C, MATRANGA C, et al. The Drosophila RNA methyltransferase, DmHen1, modifies germline piRNAs and singlestranded siRNAs in RISC [J]. Curr Biol, 2007, 17(14): 1265-1272.
[26] SAITO K, SAKAGUCHI Y, SUZUKI T, et al. Pimet, the Drosophila homolog of HEN1, mediates 2'-O-methylation of Piwi-interacting RNAs at their 3' ends [J]. Genes Dev, 2007, 21(13): 1603-1608.
[27] OHARA T, SAKAGUCHI Y, SUZUKI T, et al. The 3' termini of mouse Piwi-interacting RNAs are 2'-O-methylated [J]. Nat Struct Mol Biol, 2007, 14(4): 349-350.
[28] KIRINO Y, MOURELATOS Z. Mouse Piwi-interacting RNAs are 2'-O-methylated at their 3' termini [J]. Nat Struct Mol Biol, 2007, 14(4): 347-348.
[29] GAINETDINOV I, SKVORTSOVA Y, KONDRATIEVA S, et al. Two modes of targeting transposable elements by piRNA pathway in human testis [J]. RNA, 2017, 23(11): 1614-1625.
[30] ZHANG F, WANG J, XU J, et al. UAP56 couples piRNA clusters to the perinuclear transposon silencing machinery [J]. Cell, 2012, 151(4): 871-884.
[31] DE FAZIO S, BARTONICEK N, DI GIACOMOM, et al. The endonuclease activity of Mili fuels piRNA amplification that silences LINE1 elements [J]. Nature, 2011, 480(7376): 259-263.
[32] DARRICARRERE N, LIU N, WATANABE T, et al. Function of Piwi, a nuclear Piwi/Argonaute protein, is independent of its slicer activity [J]. Proc Natl Acad Sci USA, 2013, 110(4): 1297-1302.
[33] FAULKNER G J, KIMURA Y, DAUB C O, et al. The regulated retrotransposon transcriptome of mammalian cells [J]. Nat Genet, 2009, 41(5): 563-571.
[34] WATANABE T, CHENG E C, ZHONG M, et al. Retrotransposons and pseudogenes regulate mRNAs and lncRNAs via the piRNA pathway in the germline [J]. Genome Res, 2015, 25(3): 368-380.
[35] VOUREKAS A, ALEXIOU P, VRETTOS N, et al. Sequencedependent but not sequence-specific piRNA adhesion traps mRNAs to the germ plasm [J]. Nature, 2016, 531(7594): 390-394.
[36] GOU L T, DAI P, YANG J H, et al. Pachytene piRNAs instruct massive mRNA elimination during late spermiogenesis [J]. Cell Res, 2014, 24(6): 680-700.
[37] SHEN E Z, CHEN H, OZTURK A R, et al. Identification of piRNA binding sites reveals the Argonaute regulatory landscape of the C. elegans germline [J]. Cell, 172(5): 937-951.
[38] ZHANG D, TU S, STUBNA M, et al. The piRNA targeting rules and the resistance to piRNA silencing in endogenous genes [J]. Science, 2018, 359(6375): 587-592.
[39] SHPIZ S, RYAZANSKY S, OLOVNIKOV I, et al. Euchromatic transposon insertions trigger production of novel Pi- and endosiRNAs at the target sites in the Drosophila germline [J]. PLoS Genet, 2014, 10(2): e1004138.
[40] KLEIN J D, QU C, YANG X, et al. c-Fos repression by Piwi regulates Drosophila ovarian germline formation and tissue morphogenesis [J]. PLoS Genet, 2016, 12(9): e1006281.
[41] BARCKMANN B, PIERSON S, DUFOURT J, et al. Aubergine iCLIP reveals piRNA-dependent decay of mRNAs involved in germ cell development in the early embryo [J]. Cell Rep, 2015, 12(7): 1205-1216.
[42] ROUGET C, PAPIN C, BOUREUX A, et al. Maternal mRNA deadenylation and decay by the piRNA pathway in the early Drosophila embryo [J]. Nature, 2010, 467(7319): 1128-1132.
[43] LIM A K, TAO L, KAI T. piRNAs mediate posttranscriptional retroelement silencing and localization to pi-bodies in the Drosophila germline [J]. J Cell Biol, 2009, 186(3): 333-342.
[44] HARRIS A N, MACDONALD P M. Aubergine encodes a Drosophila polar granule component required for pole cell formation and related to eIF2C [J]. Development, 2001, 128(14): 2823-2832.
[45] RAMAT A, GARCIA-SILVA M, JAHAN C, et al. The PIWI protein Aubergine recruits eIF3 to activate translation in the germ plasm [J]. Cell Res, 2020, 30(5): 421-435.
[46] DAI P, WANG X, GOU L-T, et al. A translation-activating function of MIWI/piRNA during mouse spermiogenesis [J]. Cell, 2019, 179(7): 1566-1581.
[47] UNHAVAITHAYA Y, HAO Y, BEYRET E, et al. MILI, a PIWIinteracting RNA-binding protein, is required for germ line stem cell self-renewal and appears to positively regulate translation [J]. J Biol Chem, 2009, 284(10): 6507-6519.
[48] GRIVNA S T, PYHTILA B, LIN H. MIWI associates with translational machinery and PIWI-interacting RNAs (piRNAs) in regulating spermatogenesis [J]. Proc Natl Acad Sci USA, 2006, 103(36): 13415-13420.
[49] DUFOURT J, BONTONOU G, CHARTIER A, et al. piRNAs and Aubergine cooperate with Wispy poly(A) polymerase to stabilize mRNAs in the germ plasm [J]. Nat Commun, 2017, 8(1): 1305.
[50] HSIEH C L, XIA J, LIN H. MIWI prevents aneuploidy during meiosis by cleaving excess satellite RNA [J]. EMBO J, 2020, 39(16): e103614. [51] QIAO D, ZEEMAN A M, DENG W, et al. Molecular characterization of hiwi, a human member of the piwi gene family whose overexpression is correlated to seminomas [J]. Oncogene, 2002, 21(25): 3988-3999.
[52] DONG P, XIONG Y, KONNO Y, et al. Critical roles of PIWIL1 in human tumors: expression, functions, mechanisms, and potential clinical implications [J]. Front Cell Dev Biol, 2021, 9: 656993.
[53] LIU Y, DOU Y, SONG X, et al. The emerging role of the piRNA/ piwi complex in cancer [J]. Mol Cancer, 2019, 18(1): 123.
[54] WANG Q E, HAN C, MILUM K, et al. Stem cell protein Piwil2 modulates chromatin modifications upon cisplatin treatment [J]. Mutat Res, 2011, 708(1/2): 59-68.
[55] LEE J H, SCHÜTTE D, WULF G, et al. Stem-cell protein Piwil2 is widely expressed in tumors and inhibits apoptosis through activation of Stat3/Bcl-XL pathway [J]. Hum Mol Genet, 2006, 15(2): 201-211.
[56] LI W, MARTINEZ-USEROS J, GARCIA-CARBONERO N, et al. The clinical significance of PIWIL3 and PIWIL4 expression in pancreatic cancer [J]. J Clin Med, 2020, 9(5): 1252.
[57] WANG Z, LIU N, SHI S, et al. The role of PIWIL4, an Argonaute family protein, in breast cancer [J]. J Biol Chem, 2016, 291(20): 10646-10658.
[58] SU C, REN Z, WANG F, et al. PIWIL4 regulates cervical cancer cell line growth and is involved in down-regulating the expression of p14ARF and p53 [J]. FEBS Lett, 2012, 586(9): 1356-1362.
[59] ZENG G, ZHANG D, LIU X, et al. Co-expression of Piwil2/Piwil4 in nucleus indicates poor prognosis of hepatocellular carcinoma [J]. Oncotarget, 2017, 8(3): 4607-4617.
[60] TAN L, MAI D, ZHANG B, et al. PIWI-interacting RNA-36712 restrains breast cancer progression and chemoresistance by interaction with SEPW1 pseudogene SEPW1P RNA [J]. Mol Cancer, 2019, 18(1): 9.
[61] SULEMAN M, CHEN A, MA H, et al. PIR promotes tumorigenesis of breast cancer by upregulating cell cycle activator E2F1 [J]. Cell Cycle, 2019, 18(21): 2914-2927.
[62] ZHANG H, REN Y, XU H, et al. The expression of stem cell protein Piwil2 and piR-932 in breast cancer [J]. Surg Oncol, 2013, 22(4): 217-223.
[63] FU A, JACOBS D I, HOFFMAN A E, et al. PIWI-interacting RNA 021285 is involved in breast tumorigenesis possibly by remodeling the cancer epigenome [J]. Carcinogenesis, 2015, 36(10): 1094-1102.
[64] REEVES M E, FIREK M, JLIEDI A, et al. Identification and characterization of RASSF1C piRNA target genes in lung cancer cells [J]. Oncotarget, 2017, 8(21): 34268-34282.
[65] LI D, LUO Y, GAO Y, et al. piR-651 promotes tumor formation in non-small cell lung carcinoma through the upregulation of cyclin D1 and CDK4 [J]. Int J Mol Med, 2016, 38(3): 927-936.
[66] YAO J, WANG Y W, FANG B B, et al. piR-651 and its function in 95-D lung cancer cells [J]. Biomed Rep, 2016, 4(5): 546-550. [67] PENG L, SONG L, LIU C, et al. piR-55490 inhibits the growth of lung carcinoma by suppressing mTOR signaling [J]. Tumour Biol, 2016, 37(2): 2749-2756.
[68] LIU X, SUN Y, GUO J, et al. Expression of hiwi gene in human gastric cancer was associated with proliferation of cancer cells [J]. Int J Cancer, 2006, 118(8): 1922-1929.
[69] CHENG J, GUO J M, XIAO B-X, et al. piRNA, the new non-coding RNA, is aberrantly expressed in human cancer cells [J]. Clin Chim Acta, 2011, 412(17/18): 1621-1625.
[70] MARTINEZ V D, ENFIELD K S S, ROWBOTHAM D A, et al. An atlas of gastric PIWI-interacting RNA transcriptomes and their utility for identifying signatures of gastric cancer recurrence [J]. Gastric Cancer, 2016, 19(2): 660-665.
[71] WENG W, LIU N, TOIYAMA Y, et al. Novel evidence for a PIWIinteracting RNA (piRNA) as an oncogenic mediator of disease progression, and a potential prognostic biomarker in colorectal cancer [J]. Mol Cancer, 2018, 17(1): 16.
[72] YIN J, JIANG X Y, QI W, et al. piR-823 contributes to colorectal tumorigenesis by enhancing the transcriptional activity of HSF1 [J]. Cancer Sci, 2017, 108(9): 1746-1756.
[73] MAI D, DING P, TAN L, et al. PIWI-interacting RNA-54265 is oncogenic and a potential therapeutic target in colorectal adenocarcinoma [J]. Theranostics, 2018, 8(19): 5213-5230.
[74] YU Y, XIAO J, HANN S S. The emerging roles of PIWI-interacting RNA in human cancers [J]. Cancer Manag Res, 2019, 11: 5895- 5909.
[75] TOSAR J P, ROVIRA C, CAYOTA A. Non-coding RNA fragments account for the majority of annotated piRNAs expressed in somatic non-gonadal tissues [J]. Commun Biol, 2018, 1: 2.
[76] SUZUKI R, HONDA S, KIRINO Y. PIWI expression and function in cancer [J]. Front Genet, 2012, 3: 204.
[77] LI F, YUAN P, RAO M, et al. piRNA-independent function of PIWIL1 as a co-activator for anaphase promoting complex/ cyclosome to drive pancreatic cancer metastasis [J]. Nat Cell Biol, 2020, 22(4): 425-438.
[78] SHI S, YANG Z Z, LIU S, et al. PIWIL1 promotes gastric cancer via a piRNA-independent mechanism [J]. Proc Natl Acad Sci USA, 2020, 117(36): 22390-22401.
[79] YAMAGUCHI S, OE A, NISHIDA K M, et al. Crystal structure of Drosophila Piwi [J]. Nat Commun, 2020, 11(1): 858.
[80] HAYASHI R, SCHNABL J, HANDLER D, et al. Genetic and mechanistic diversity of piRNA 3'-end formation [J]. Nature, 2016, 539(7630): 588-592.
[81] WANG H, MA Z, NIU K, et al. Antagonistic roles of Nibbler and Hen1 in modulating piRNA 3' ends in Drosophila [J]. Development, 2016, 143(3): 530-539.
[82] SAITO K, INAGAKI S, MITUYAMA T, et al. A regulatory circuit for piwi by the large Maf gene traffic jam in Drosophila [J]. Nature, 2009, 461(7268): 1296-1299.
/
| 〈 |
|
〉 |