

Development and application of C2H2 zinc finger protein-based drug delivery technology
Received date: 2021-06-19
Online published: 2021-10-22
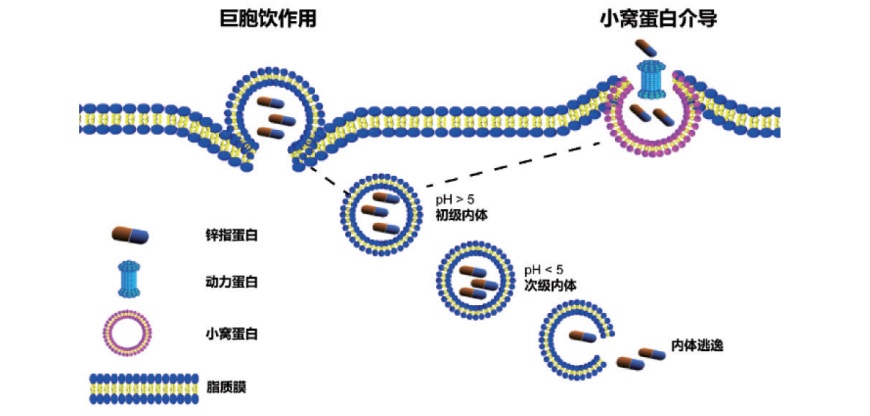
LI Lu, WEI Xuan, LIU Jia . Development and application of C2H2 zinc finger protein-based drug delivery technology[J]. Chinese Journal of Nature, 2021 , 43(5) : 359 -364 . DOI: 10.3969/j.issn.0253-9608.2021.05.006
[1] VALLEE B L, FALCHUK K H. The biochemical basis of zinc physiology [J]. Physiol Rev, 1993, 73: 79-118.
[2] FRANKEL A D, PABO C O. Fingering too many proteins [J]. Cell, 1988, 53: 675.
[3] BERG J M, SHI Y. The galvanization of biology: a growing appreciation for the roles of zinc [J]. Science, 1996, 271: 1081- 1085.
[4] HARRISON S C. A structural taxonomy of DNA-binding domains [J]. Nature, 1991, 353: 715-719.
[5] COX D B, PLATT R J, ZHANG F. Therapeutic genome editing: prospects and challenges [J]. Nat Med, 2015, 21: 121-131.
[6] KIM Y G, CHA J, CHANDRASEGARAN S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain [J]. PNAS, 1996, 93: 1156-1160.
[7] GERSBACH C A, GAJ T, BARBAS C F, et al. Synthetic zinc finger proteins: the advent of targeted gene regulation and genome modification technologies [J]. Acc Chem Res, 2014, 47: 2309-2318.
[8] TEBAS P, STEIN D, TANG W W. et al. Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV [J]. N Engl J Med, 2014, 370: 901-910.
[9] GAJ T, GUO J, KATO Y. et al. Targeted gene knockout by direct delivery of zinc-finger nuclease proteins [J]. Nat Methods, 2012, 9: 805-807.
[10] LIU J, GAJ T, PATTERSON J T, et al. Cell-penetrating peptidemediated delivery of TALEN proteins via bioconjugation for genome engineering [J]. PLoS One, 2014, 9: e85755.
[11] KIM S, KIM D, CHO S W, et al. Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins [J]. Genome Res, 2014, 24: 1012-1019.
[12] GAJ T, LIU J, ANDERSON K E, et al. Protein delivery using Cys2- His2 zinc-finger domains [J]. ACS Chem Bio, 2014, 9: 1662-1667.
[13] LIU J, SHUI S L. Delivery methods for site-specific nucleases: Achieving the full potential of therapeutic gene editing [J]. J Control Release, 2016, 244: 83-97.
[14] GILLMEISTER M P, BETENBAUGH M J, FISHMAN P S. Cellular trafficking and photochemical internalization of cell penetrating peptide linked cargo proteins: a dual fluorescent labeling study [J]. Bioconjug Chem, 2011, 22: 556-566.
[15] MAI J C, SHEN H, WATKINS S C, et al. Efficiency of protein transduction is cell type-dependent and is enhanced by dextran sulfate [J]. Biochem J, 2002, 277: 30208-30218.
[16] LINDSAY M A. Peptide-mediated cell delivery: application in protein target validation [J]. Curr Opin Biotechnol, 2002, 2: 587- 594.
[17] VANDENBERG A, DOWDY S F. Protein transduction domain delivery of therapeutic macromolecules [J]. Curr Opin Biotechnol, 2011, 22: 888-893.
[18] KOREN E, TORCHILIN V P. Cell-penetrating peptides: breaking through to the other side [J]. Trends Mol Med, 2012, 18: 385-393.
[19] LIU J, GAJ T, WALLEN M C, et al. Improved cell-penetrating zincfinger nuclease proteins for precision genome engineering [J]. Mol Ther Nucleic Acids, 2015, 4: e232.
[20] PANG X, TONG Y, LI F, et al. Expression and characterization of human lactoferrin with tandem zinc finger protein in Chlamydomonas reinhardtii [J]. Algal Res, 2019, 43: 101635.
[21] SUZUKI Y A, LOPEZ V, LONNERDAL B. Mammalian lactoferrin receptors: structure and function [J]. Cell Mol Life Sci, 2005, 62: 2560-2575.
[22] SALVEMINI D, RILEY D P, CUZZOCREA S. SOD mimetics are coming of age [J]. Nat Rev Drug Discov, 2002, 1: 367-374.
[23] FRIDOVICH I. Superoxide radical and superoxide dismutases [J]. Annu Rev Biochem, 1995, 64: 97-112.
[24] MCCORD J M. Free radicals and inflammation: protection of synovial fluid by superoxide dismutase [J]. Science, 1974, 185: 529- 531.
[25] YANG G, CHAN P H, CHEN J, et al. Human copper-zinc superoxide dismutase transgenic mice are highly resistant to reperfusion injury after focal cerebral ischemia [J]. Stroke, 1994, 25: 165-170.
[26] LAND W, ZWELER J L. Prevention of reperfusion-induced, free radical-mediated acute endothelial injury by superoxide dismutase as an effective tool to delay/prevent chronic renal allograft failure: a review [J]. Transplant Proc, 1997, 29: 2567-2568.
[27] SHINGU M, TAKAHASHI S, ITO M, et al. Anti-inflammatory effects of recombinant human manganese superoxide dismutase on adjuvant arthritis in rats [J]. Rheumatol Int, 1994, 14: 77-81.
[28] LAZZARI F D, BUBACCO L, WHITWORTH A J, et al. Superoxide radical dismutation as new therapeutic strategy in parkinson’s disease [J]. Aging Dis, 2018, 9: 716-728.
[29] SAFFORD S E, OBERLEY T D, URANO M, et al. Suppression of fibrosarcoma metastasis by elevated expression of manganese superoxide dismutase [J]. Cancer Res, 1994, 54: 4261-4265.
[30] CHURCH S L, GRANT J W, RIDNOUR L A, et al, Increased manganese superoxide dismutase expression suppresses the malignant phenotype of human melanoma cells [J]. PNAS, 1993, 90: 3113-3117.
[31] MOLLACE V, NOTTET H S, CLAYETTE P, et al. Oxidative stress and neuroAIDS: triggers, modulators and novel antioxidants [J]. Trends Neurosci, 2001, 24: 411-416. [32] LIU J, LI J, LI J, et al. Delivery of superoxide dismutase using Cys2- His2 zinc-finger proteins [J]. Methods Mol Biol, 2018, 1867: 113- 123.
/
| 〈 |
|
〉 |