

The mRNA technology won the 2023 Nobel Prize in Physiology or Medicine
Received date: 2023-10-25
Online published: 2023-12-21
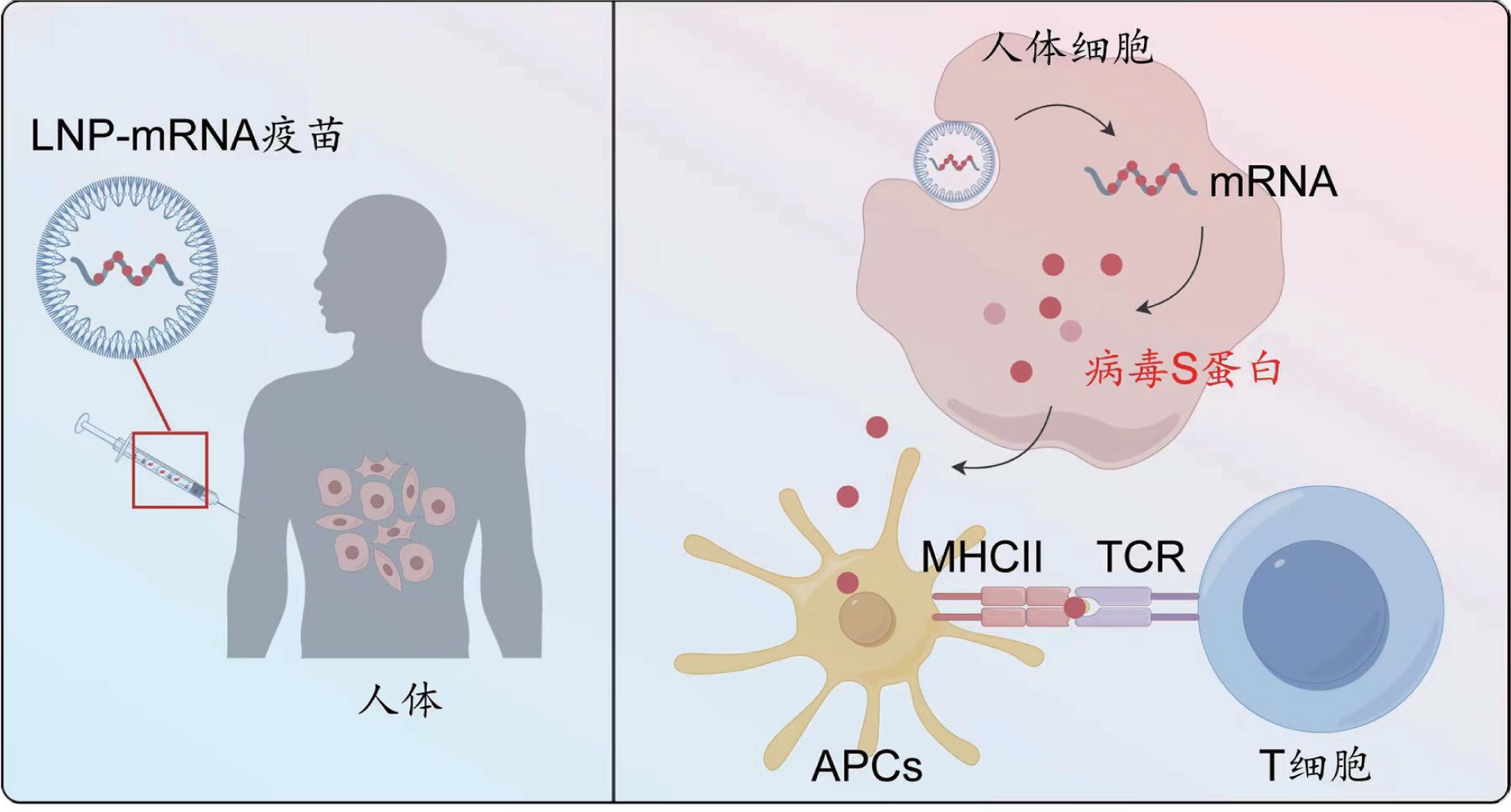
XU Yao, LI Kexin, ZHAO Lianmei, ZHANG Hongtao . The mRNA technology won the 2023 Nobel Prize in Physiology or Medicine[J]. Chinese Journal of Nature, 2023 , 45(6) : 423 -428 . DOI: 10.3969/j.issn.0253-9608.2023.06.004
[J]. Nature, 1961, 190: 576-581.
[2] WATANABE K, YOKOBORI S. Genetic code: introduction [EB/OL]. (2011-10-17)[2023-10-25]. https://onlinelibrary.wiley.com/
doi/10.1002/9780470015902.a0000809.pub2.
[3] MELTON D A, KRIEG P A, REBAGLIATI M R, et al. Efficient in vitro synthesis of biologically active RNA and RNA hybridization
probes from plasmids containing a bacteriophage SP6 promoter [J].Nucleic Acids Res, 1984, 12(18): 7035-7056.
[4] KARIKÓ K, BUCKSTEIN M, NI H, et al. Suppression of RNA recognition by toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA [J]. Immunity, 2005, 23(2): 165-175.
[5] KARIKÓ K, KUO A, BARNATHAN E S, et al. Phosphateenhanced transfection of cationic lipid-complexed mRNA and plasmid DNA [J]. Biochim Biophys Acta, 1998, 1369(2): 320-334.
[6] ROJAS L A, SETHNA Z, SOARES K C, et al. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer [J].
Nature, 2023, 618(7963): 144-150.
[7] MACKENSEN A, HAANEN J B A G, KOENECKE C, et al. CLDN6-specific CAR-T cells plus amplifying RNA vaccine in
relapsed or refractory solid tumors: the phase 1 BNT211-01 trial [J]. Nat Med, 2023, 29(11): 2844-2853.
[8] QIU M, GLASS Z, CHEN J, et al. Lipid nanoparticle-mediated codelivery of Cas9 mRNA and single-guide RNA achieves liverspecific
in vivo genome editing of Angptl3 [J]. Proc Natl Acad Sci USA, 2021, 118(10): e2020401118.
[9] RURIK J G, TOMBÁCZ I, YADEGARI A, et al. CAR T cells produced in vivo to treat cardiac injury [J]. Science, 2022, 375(6576): 91-96.
[10] CHENG Q, WEI T, FARBIAK L, et al. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and
CRISPR–Cas gene editing [J]. Nat Nanotechnol, 2020, 15(4): 313-320.
[11] DOLGIN E. The tangled history of mRNA vaccines [J]. Nature, 2021, 597: 318-324.
/
| 〈 |
|
〉 |