

Research progress in targeted degradation of membrane proteins
Received date: 2022-12-30
Online published: 2023-12-21
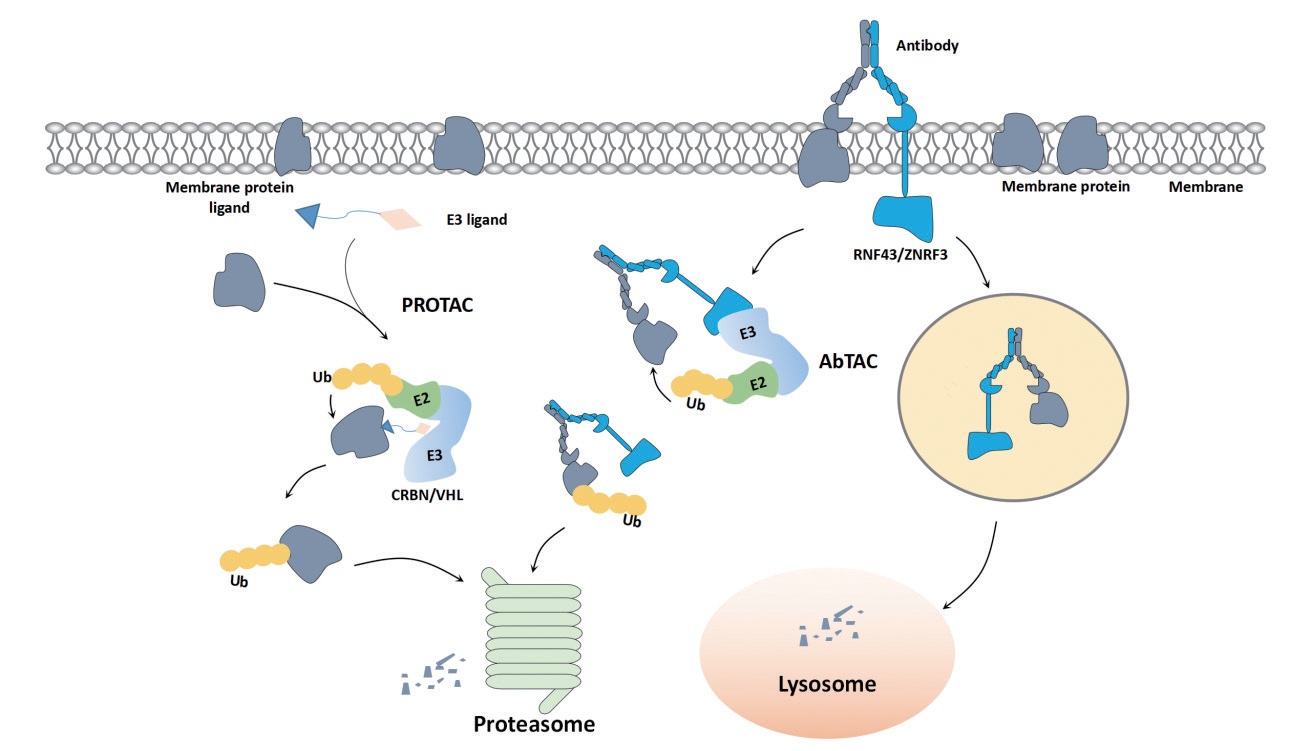
ZHAO Su, ZHANG Heng, LI Yan, WANG Zhenchao . Research progress in targeted degradation of membrane proteins[J]. Chinese Journal of Nature, 2023 , 45(6) : 434 -442 . DOI: 10.3969/j.issn.0253-9608.2023.06.008
[9] BONDESON D P, MARES A, SMITH I E, et al. Catalytic in vivo protein knockdown by small-molecule PROTACs [J]. Nat Chem
Biol, 2015, 11(8): 611-617.
[10] MULLARD A. Targeted protein degraders crowd into the clinic [J]. Nat Rev Drug Discov, 2021, 20(4): 247-250.
[11] BANIK S M, PEDRAM K, WISNOVSKY S, et al. Lysosometargeting chimaeras for degradation of extracellular proteins [J]. Nature, 2020, 584(7820): 291-297.
[12] COTTON A D, NGUYEN D P, GRAMESPACHER J A, et al. Development of antibody-based PROTACs for the degradation of
the cell-surface immune checkpoint protein PD-L1 [J]. J Am Chem Soc, 2021, 143(2): 593-598.
[13] ZHANG H, HAN Y, YANG Y, et al. Covalently engineered nanobody chimeras for targeted membrane protein degradation [J]. J
Am Chem Soc, 2021, 143(40): 16377-16382.
[14] PANCE K, GRAMESPACHER J A, BYRNES J R, et al. Modular cytokine receptor-targeting chimeras for targeted degradation of cell surface and extracellular proteins [J]. Nat Biotechnol, 2023, 41(2): 273-281.
[15] SAKAMOTO K M, KIM K B, KUMAGAI A, et al. Protacs: chimeric molecules that target proteins to the Skp1-Cullin-F box complex for ubiquitination and degradation [J]. Proc Natl Acad Sci USA, 2001, 98(15): 8554-8559.
[16] ZHANG C, HAN X R, YANG X, et al. Proteolysis targeting chimeras (PROTACs) of anaplastic lymphoma kinase (ALK) [J].
Eur J Med Chem, 2018, 151: 304-314.
[17] QU X, LIU H, SONG X, et al. Effective degradation of EGFRL858R+T790M mutant proteins by CRBN-based PROTACs through
both proteosome and autophagy/lysosome degradation system [J]. Eur J Med Chem, 2021, 218: 113328.
[18] ALABI S, JAIME F S, YAO Z, et al. Mutant-selective degradation by BRAF-targeting PROTACs [J]. Nat Commun, 2021, 12(1): 920.
[19] DIEHL C J, CIULLI A. Discovery of small molecule ligands for the von Hippel-Lindau (VHL) E3 ligase and their use as inhibitors and PROTAC degraders [J]. Chem Soc Rev, 2022, 51(19): 8216-8257.
[20] COUTINHO M F, PRATA M J, ALVES S. A shortcut to the lysosome: the mannose-6-phosphate-independent pathway [J]. Mol
Genet Metab, 2012, 107(3): 257-266.
[21] AHN G, BANIK S M, MILLER C L, et al. LYTACs that engage theasialo glycoprotein receptor for targeted protein degradation [J]. Nat Chem Biol, 2021, 17(9): 937-946.
[22] CAIANIELLO D F, ZHANG M, RAY J D, et al. Bifunctional small molecules that mediate the degradation of extracellular proteins [J].
Nat Chem Biol, 2021, 17(9): 947-953.
[23] MAREI H, TSAI W T K, KEE Y S, et al. Antibody targeting of E3 ubiquitin ligases for receptor degradation [J]. Nature, 2022,
610(7930): 182-189.
[24] JANSSENS R, STRUYF S, PROOST P. The unique structural and functional features of CXCL12 [J]. Cell Mol Immunol, 2018, 15(4):
299-311.
[25] CHIN J W. Expanding and reprogramming the genetic code [J]. Nature, 2017, 550(7674): 53-60.
[26] UHLÉN M, FAGERBERG L, HALLSTRÖM B M, et al. Tissuebased map of the human proteome [J]. Science, 2015, 347(6220):
1260419.
[27] CHAMBERLAIN P P, HAMANN L G. Development of targeted protein degradation therapeutics [J]. Nat Chem Biol, 2019, 15(10): 937-944.
/
| 〈 |
|
〉 |