

Developing the affinity mass spectrometry technology for the discovery of chemical ligands and drug leads towards GPCR targets
Received date: 2020-10-01
Online published: 2021-02-25
ZHANG Bingjie, SHUI Wenqing . Developing the affinity mass spectrometry technology for the discovery of chemical ligands and drug leads towards GPCR targets[J]. Chinese Journal of Nature, 2021 , 43(1) : 32 -38 . DOI: 10.3969/j.issn.0253-9608.2021.01.005
[1] SANTOS R, URSU O, GAULTON A, et al. A comprehensive map of molecular drug targets [J]. Nat Rev Drug Discov, 2017, 16(1): 19-34.
[2] GOODNOW JR R A, DUMELIN C E, KEEFE A D. DNA-encoded chemistry: enabling the deeper sampling of chemical space [J]. Nat Rev Drug Discov, 2017, 16(2): 131-147.
[3] KLEINER R E, DUMELIN C ELIU D R. Small-molecule discovery from DNA-encoded chemical libraries [J]. Chem Soc Rev, 2011, 40(12): 5707-5717.
[4] KODADEK T, PACIARONI N G, BALZARINI M, et al. Beyond protein binding: Recent advances in screening DNA-encoded libraries [J]. Chem Commun (Camb), 2019, 55(89): 13330-13341.
[5] NERI D, LERNER R A. DNA-encoded chemical libraries: A selection system based on endowing organic compounds with amplifiable information [J]. Annu Rev Biochem, 2018, 87: 479-502.
[6] AHN S, KAHSAI A W, PANI B, et al. Allosteric “beta-blocker” isolated from a DNA-encoded small molecule library [J]. Proc Natl Acad Sci USA, 2017, 114(7): 1708-1713.
[7] AHN S, PANI B, KAHSAI A W, et al. Small-molecule positive allosteric modulators of the beta2-adrenoceptor isolated from DNAencoded libraries [J]. Mol Pharmacol, 2018, 94(2): 850-861.
[8] WU Z, GRAYBILL T L, ZENG X, et al. Cell-based selection expands the utility of DNA-encoded small-molecule library technology to cell surface drug targets: Identification of novel antagonists of the NK3 tachykinin receptor [J]. ACS Comb Sci, 2015, 17(12): 722-731.
[9] ANNIS A, CHUANG C C, NAZEF N. Mass spectrometry in medicinal chemistry [M]. Weinheim, Germany: WILEY -VCH Verlag GmbH & Co KGaA, 2007: 121-156.
[10] CHEN X, LI L, CHEN S, et al. Identification of inhibitors of the antibiotic-resistance target New Delhi metallo-beta-lactamase 1 by both nanoelectrospray ionization mass spectrometry and ultrafiltration liquid chromatography/mass spectrometry approaches [J]. Anal Chem, 2013, 85(16): 7957-7965.
[11] CHEN X, QIN S, CHEN S, et al. A ligand-observed mass spectrometry approach integrated into the fragment based lead discovery pipeline [J]. Sci Rep, 2015, 5: 8361.
[12] GESMUNDO N J, SAUVAGNAT B, CURRAN P J, et al. Nanoscale synthesis and affinity ranking [J]. Nature, 2018, 557(7704): 228-232.
[13] O’CONNELL T N, RAMSAY J, RIETH S F, et al. Solution-based indirect affinity selection mass spectrometry—a general tool for high-throughput screening of pharmaceutical compound libraries [J]. Anal Chem, 2014, 86(15): 7413-7420.
[14] QIN S, REN Y, FU X, et al. Multiple ligand detection and affinity measurement by ultrafiltration and mass spectrometry analysis applied to fragment mixture screening [J]. Anal Chim Acta, 2015, 886: 98-106.
[15] KUMARI P, GHOSH E, SHUKLA A K. Emerging approaches to GPCR ligand screening for drug discovery [J]. Trends Mol Med, 2015, 21(11): 687-701.
[16] CALLERI E, CERUTI S, CRISTALLI G, et al. Frontal affinity chromatography-mass spectrometry useful for characterization of new ligands for GPR17 receptor [J]. J Med Chem, 2010, 53(9): 3489-3501.
[17] MA J, LU Y, WU D, et al. Ligand identification of the adenosine A2A receptor in self-assembled nanodiscs by affinity mass spectrometry [J]. Anal Methods, 2017, 9(40): 5851-5858.
[18] MASSINK A, HOLZHEIMER M, HOLSCHER A, et al. Mass spectrometry-based ligand binding assays on adenosine A1 and A2A receptors [J]. Purinergic Signal, 2015, 11(4): 581-594.
[19] QIN S, MENG M, YANG D, et al. High-throughput identification of G protein-coupled receptor modulators through affinity mass spectrometry screening [J]. Chem Sci, 2018, 9(12): 3192-3199.
[20] TEMPORINI C, MASSOLINI G, MARUCCI G, et al. Development of new chromatographic tools based on A2A adenosine receptor subtype for ligand characterization and screening by FAC-MS [J]. Anal Bioanal Chem, 2013, 405(2/3): 837-845.
[21] WHITEHURST C E, YAO Z, MURPHY D, et al. Application of affinity selection-mass spectrometry assays to purification and affinity-based screening of the chemokine receptor CXCR4 [J]. Comb Chem High Throughput Screen, 2012, 15(6): 473-485.
[22] YEN H Y, HOI K K, LIKO I, et al. PtdIns(4,5)P2 stabilizes active states of GPCRs and enhances selectivity of G-protein coupling [J]. Nature, 2018, 559(7714): 423-427.
[23] YEN H Y, HOPPER J T S, LIKO I, et al. Ligand binding to a G protein-coupled receptor captured in a mass spectrometer [J]. Sci Adv, 2017, 3(6): e1701016.
[24] DENG Y, SHIPPS JR G W, COOPER A, et al. Discovery of novel, dual mechanism ERK inhibitors by affinity selection screening of an inactive kinase [J]. J Med Chem, 2014, 57(21): 8817-8826.
[25] KUTILEK V D, ANDREWS C L, RICHARDS M P, et al. Integration of affinity selection-mass spectrometry and functional cell-based assays to rapidly triage druggable target space within the NF-κB pathway [J]. J Biomol Screen, 2016, 21(6): 608-619.
[26] WALKER S S, DEGEN D, NICKBARG E, et al. Affinity selectionmass spectrometry identifies a novel antibacterial RNA polymerase inhibitor [J]. ACS Chem Biol, 2017, 12(5): 1346-1352.
[27] ZHANG T, LIU Y, YANG X, et al. Definitive metabolite identification coupled with automated ligand identification system (ALIS) technology: A novel approach to uncover structure-activity relationships and guide drug design in a factor IXa inhibitor program [J]. J Med Chem, 2016, 59(5): 1818-1829.
[28] LU Y, QIN S, ZHANG B, et al. Accelerating the throughput of affinity mass spectrometry-based ligand screening toward a G protein-coupled receptor [J]. Anal Chem, 2019, 91(13): 8162-8169.
[29] CHOI Y, JERMIHOV K, NAM S J, et al. Screening natural products for inhibitors of quinone reductase-2 using ultrafiltration LC-MS [J]. Anal Chem, 2011, 83(3): 1048-1052.
[30] FU X, WANG Z, LI L, et al. Novel chemical ligands to Ebola virus and Marburg virus nucleoproteins identified by combining affinity mass spectrometry and metabolomics approaches [J]. Sci Rep, 2016, 6: 29680.
[31] SONG H P, CHEN J, HONG J Y, et al. A strategy for screening of high-quality enzyme inhibitors from herbal medicines based on ultrafiltration LC-MS and in silico molecular docking [J]. Chem Commun (Camb), 2015, 51(8): 1494-1497.
[32] WANG L, LIU Y, LUO Y, et al. Quickly screening for potential alpha-glucosidase inhibitors from guava leaves tea by bioaffinity ultrafiltration coupled with HPLC-ESI-TOF/MS method [J]. J Agric Food Chem, 2018, 66(6): 1576-1582.
[33] WANG Z, LIANG H, CAO H, et al. Efficient ligand discovery from natural herbs by integrating virtual screening, affinity massspectrometry and targeted metabolomics [J]. Analyst, 2019, 144(9): 2881-2890.
[34] YANG Z, ZHANG Y, SUN L, et al. An ultrafiltration highperformance liquid chromatography coupled with diode array detector and mass spectrometry approach for screening and characterising tyrosinase inhibitors from mulberry leaves [J]. Anal Chim Acta, 2012, 719: 87-95.
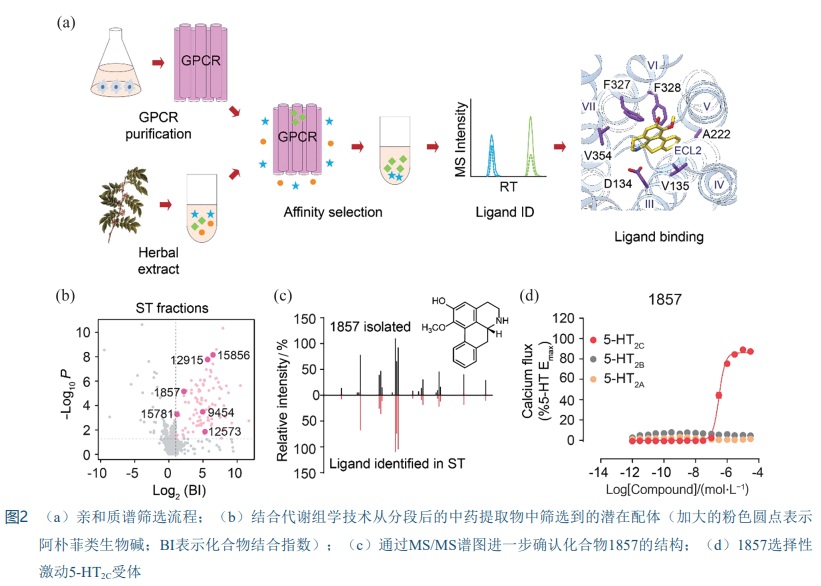
[35] ZHANG B, ZHAO S, YANG D, et al. A Novel G protein-biased and subtype-selective agonist for a G protein-coupled receptor discovered from screening herbal extracts [J]. ACS Cent Sci, 2020, 6(2): 213-225.
[36] UDUGAMASOORIYA D G, DINEEN S P, BREKKEN R A, et al. A peptoid “antibody surrogate” that antagonizes VEGF receptor 2 activity [J]. J Am Chem Soc, 2008, 130(17): 5744-5752.
[37] HOFNER G, WANNER K T. Competitive binding assays made easy with a native marker and mass spectrometric quantification [J]. Angew Chem Int Ed Engl, 2003, 42(42): 5235-5237.
[38] NIESSEN K V, HOFNER G, WANNER K T. Competitive MS binding assays for dopamine D2 receptors employing spiperone as a native marker [J]. ChemBioChem, 2005, 6(10): 1769-1775.
[39] ZEPPERITZ C, HOFNER G, WANNER K T. MS-binding assays: kinetic, saturation, and competitive experiments based on quantitation of bound marker as exemplified by the GABA transporter mGAT1 [J]. ChemMedChem, 2006, 1(2): 208-217.
[40] NEIENS P, HOFNER G, WANNER K T. MS binding assays for D1 and D5 dopamine receptors [J]. ChemMedChem, 2015, 10(11): 1924-1931.
[41] SCHULLER M, HOFNER G, WANNER K T. Simultaneous multiple MS binding assays addressing D1 and D2 dopamine receptors [J]. ChemMedChem, 2017, 12(19): 1585-1594.
/
| 〈 |
|
〉 |