

Applications of optical tweezers and DNA nanotechnology in membrane biology
Received date: 2022-06-24
Online published: 2023-02-10
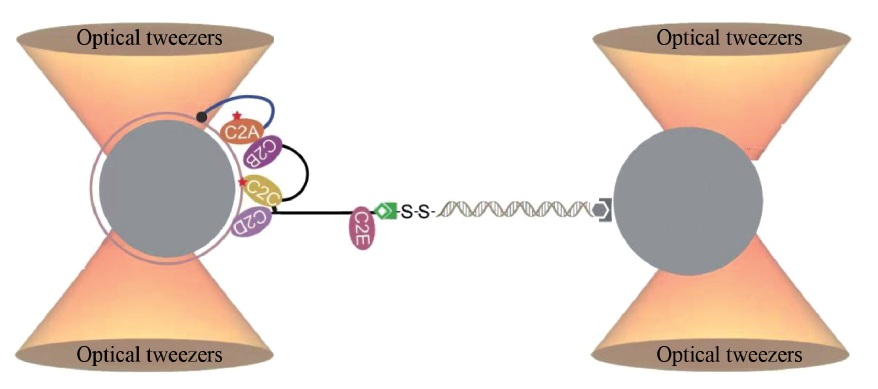
Biological membranes are platforms for signal transduction and material transport in cellular activities. In recent years, the applications of multidisciplinary approaches have shed new lights on the mechanisms of membrane protein-mediated membrane fusion and division, vesicle formation and secretion, and lipid metabolism etc. For example, the single-molecule optical tweezers, which accurately and quantitatively detect the interactions between proteins and membranes, provide a powerful approach to understand the regulatory mechanisms of such interactions at spatiotemporal level. In addition, DNA nanotechnology, which uses DNA molecules to construct programmable and self-assembled nanostructures, provides molecular devices that can be precisely modified and functionalized. The hydrophobically modified DNA nanostructures can act on phopholipid bilayers or biological membranes to modify lipid properties, modulate membrane structures, control membrane parameters and regulate transmembrane communications. The advances in these techniques will contribute to the mechanistic study of cell biology, the analysis and detection of secretory vesicles, the optimization of artificial liposome preparation, and the development of new drugs and carriers. These techniques will also provide novel systems for synthetic biology, chemical biology, and molecular medicine.
LIN Xiaona, SHI Lijun, YE Yang, WANG Yunyun, MA Lu, YANG Yang, BIAN Xin . Applications of optical tweezers and DNA nanotechnology in membrane biology[J]. Chinese Journal of Nature, 2023 , 45(1) : 45 -53 . DOI: 10.3969/j.issn.0253-9608.2023.01.005
6545.
[3] ALLEN T M, CULLIS P R. Liposomal drug delivery systems: from concept to clinical applications [J]. Adv Drug Deliv Rev, 2013, 65: 36-48.
[4] SZOSTAK J W, BARTEL D P, LUISI P L. Synthesizing life [J]. Nature, 2001, 409: 387-390.
[5] LEE Y K, LEE H, NAM J M. Lipid-nanostructure hybrids and their applications in nanobiotechnology [J]. NPG Asia Materials, 2013, 5: e48.
[6] BOGDANOV M, MILEYKOVSKAYA E, DOWHAN W. Lipids in the assembly of membrane proteins and organization of protein
supercomplexes: implications for lipid-linked disorders [J]. Subcell Biochem, 2008, 49: 197-239.
[7] ROBERTSON J L. The lipid bilayer membrane and its protein constituents [J]. J Gen Physiol, 2018, 150: 1472-1483.
[8] GE J, BIAN X, MA L, et al. Stepwise membrane binding of extended synaptotagmins revealed by optical tweezers [J]. Nat
Chem Biol, 2022, 18: 313-320.
[9] MA L, CAI Y, LI Y, et al. Single-molecule force spectroscopy of protein-membrane interactions [J]. eLife, 2017, 6: e30493.
[10] HARDY D, BILL R M, JAWHARI A, et al. Overcoming bottlenecks in the membrane protein structural biology pipeline [J]. Biochem Soc Trans, 2016, 44: 838-844.
[11] GAO Y, ZORMAN S, GUNDERSEN G, et al. Single reconstituted neuronal SNARE complexes zipper in three distinct stages [J].
Science, 2012, 337: 1340-1343.
[12] ZHANG X, MA L, ZHANG Y. High-resolution optical tweezers for single-molecule manipulation [J]. Yale J Biol Med, 2013, 86: 367-383.
[13] MOFFITT J R, CHEMLA Y R, IZHAKY D, et al. Differential detection of dual traps improves the spatial resolution of optical
tweezers [J]. Proc Natl Acad Sci USA, 2006, 103: 9006-9011.
[14] MOFFITT J R, CHEMLA Y R, SMITH S B, et al. Recent advances in optical tweezers [J]. Annu Rev Biochem, 2008, 77: 205-228.
[15] BUSTAMANTE C J, CHEMLA Y R, LIU S, et al. Optical tweezers in single-molecule biophysics [J]. Nat Rev Methods Primers, 2021, 1: 25.
[16] CHAPMAN E R. A Ca2+ sensor for exocytosis [J]. Trends Neurosci, 2018, 41: 327-330.
[17] GRUSHIN K, WANG J, COLEMAN J, et al. Structural basis for the clamping and Ca2+ activation of SNARE-mediated fusion by
synaptotagmin [J]. Nat Commun, 2019, 10: 2413.
[18] MARTENS S, KOZLOV M M, MCMAHON H T. How synaptotagmin promotes membrane fusion [J]. Science, 2007, 316: 1205-1208.
[19] PEREZ-LARA A, THAPA A, NYENHUIS S B, et al. PtdInsP2 and PtdSer cooperate to trap synaptotagmin-1 to the plasma membrane in the presence of calcium [J]. eLife, 2016, 5: e15886.
[20] SUDHOF T C. A molecular machine for neurotransmitter release: synaptotagmin and beyond [J]. Nat Med, 2013, 19: 1227-1231.
[21] MIN S-W, CHANG W-P, SÜDHOF T C. E-Syts, a family of membranous Ca2+-sensor proteins with multiple C2 domains [J].
Proc Natl Acad Sci USA, 2007, 104: 3823-3828.
[22] BIAN X, SAHEKI Y, DE CAMILLI P. Ca2+ releases E-Syt1 autoinhibition to couple ER-plasma membrane tethering with lipid
transport [J]. EMBO J, 2018, 37: 219-234.
[23] CHANG C L, HSIEH T S, YANG T T, et al. Feedback regulation of receptor-induced Ca2+ signaling mediated by E-Syt1 and Nir2 at endoplasmic reticulum-plasma membrane junctions [J]. Cell Rep, 2013, 5: 813-825.
[24] GIORDANO F, SAHEKI Y, IDEVALL-HAGREN O, et al. PI(4,5)P2-dependent and Ca2+-regulated ER-PM interactions mediated by
the extended synaptotagmins [J]. Cell, 2013, 153: 1494-1509.
[25] SAHEKI Y, BIAN X, SCHAUDER C M, et al. Control of plasma membrane lipid homeostasis by the extended synaptotagmins [J].
Nat Cell Biol, 2016, 18: 504-515.
[26] SCHAUDER C M, WU X, SAHEKI Y, et al. Structure of a lipidbound extended synaptotagmin indicates a role in lipid transfer [J].
Nature, 2014, 510: 552-555.
[27] YU H, LIU Y, GULBRANSΟN D R, et al. Extended synaptotagmins are Ca2+-dependent lipid transfer proteins at membrane contact sites [J]. Proc Natl Acad Sci USA, 2016, 113: 4362-4367.
[28] CORBALAN-GARCIA S, GÓMEZ-FERNÁNDEZ J C. Signaling through C2 domains: more than one lipid target [J]. Biochim
Biophys Acta, 2014, 1838: 1536-1547.
[29] FUSON K L, MONTES M, ROBERT J J, et al. Structure of human synaptotagmin 1 C2AB in the absence of Ca2+ reveals a novel
domain association [J]. Biochemistry, 2007, 46: 13041-13048.
[30] HURLEY J H. Membrane binding domains [J]. Biochim Biophys Acta, 2006, 1761: 805-811.
[31] ROTHEMUND P W. Folding DNA to create nanoscale shapes and patterns [J]. Nature, 2006, 440: 297-302.
[32] SEEMAN N C, SLEIMAN H F. DNA nanotechnology [J]. Nature Reviews Materials, 2018, 3: 17068.
[33] HONG F, ZHANG F, LIU Y, et al. DNA origami: scaffolds for creating higher order structures [J]. Chemical Reviews, 2017, 117:
12584-12640.
[34] DOUGLAS S M, MARBLESTONE A H, TEERAPITTAYANON S, et al. Rapid prototyping of 3D DNA-origami shapes with caDNAno
[J]. Nucleic Acids Res, 2009, 37: 5001-5006.
[35] YANG Y R, LIU Y, YAN H. DNA nanostructures as programmable biomolecular scaffolds [J]. Bioconjug Chem, 2015, 26: 1381-1395.
[36] SHEN Q, GROME M W, YANG Y, et al. Engineering lipid membranes with programmable DNA nanostructures [J]. Advanced
Biosystems, 2020, 4: 1900215.
[37] LANGECKER M, ARNAUT V, LIST J, et al. DNA nanostructures interacting with lipid bilayer membranes [J]. Accounts of Chemical Research, 2014, 47: 1807-1815.
[38] BÖRJESSON K, WIBERG J, EL-SAGHEER A H, et al. Functionalized nanostructures: redox-active porphyrin anchors for
supramolecular DNA assemblies [J]. ACS Nano, 2010, 4: 5037-5046.
[39] BÖRJESSON K, LUNDBERG E P, WOLLER J G, et al. Softsurface DNA nanotechnology: DNA constructs anchored and
aligned to lipid membrane [J]. Angewandte Chemie-International Edition, 2011, 50: 8312-8315.
[40] SUZUKI Y, ENDO M, YANG Y Y, et al. Dynamic assembly/disassembly processes of photoresponsive DNA origami
nanostructures directly visualized on a lipid membrane surface [J]. Journal of the American Chemical Society, 2014, 136: 1714-1717.
[41] LIST J, WEBER M, SIMMEL F C. Hydrophobic actuation of a DNA origami bilayer structure [J]. Angewandte Chemie-
International Edition, 2014, 53: 4236-4239.
[42] AVAKYAN N, CONWAY J W, SLEIMAN H F. Long-range ordering of blunt-ended DNA tiles on supported lipid bilayers [J]. Journal of the American Chemical Society, 2017, 139: 12027-12034.
[43] KOCABEY S, KEMPTER S, LIST J, et al. Membrane-assisted growth of DNA origami nanostructure arrays [J]. ACS Nano, 2015,
9: 3530-3539.
[44] SUZUKI Y, ENDO M, SUGIYAMA H. Lipid-bilayer-assisted twodimensional self-assembly of DNA origami nanostructures [J].
Nature Communications, 2015, 6: 8052.
[45] KHMELINSKAIA A, FRANQUELIM H G, PETROV E P, et al. Effect of anchor positioning on binding and diffusion of elongated
3D DNA nanostructures on lipid membranes [J]. Journal of Physics D-Applied Physics, 2016, 49: 194001.
[46] KHMELINSKAIA A, MUCKSCH J, PETROV E P, et al. Control of membrane binding and diffusion of cholesteryl-modified DNA
origami nanostructures by DNA spacers [J]. Langmuir, 2018, 34: 14921-14931.
[47] MALEROD L, STENMARK H. ESCRTing membrane deformation [J]. Cell, 2009, 136: 15-17.
[48] SAARIKANGAS J, ZHAO H, PYKÄLÄINEN A, et al. Molecular mechanisms of membrane deformation by I-BAR domain proteins
[J]. Curr Biol, 2009, 19: 95-107.
[49] CZOGALLA A, KAUERT D J, FRANQUELIM H G, et al. Amphipathic DNA origami nanoparticles to scaffold and deform
lipid membrane vesicles [J]. Angewandte Chemie-International Edition, 2015, 54: 6501-6505.
[50] FRANQUELIM H G, KHMELINSKAIA A, SOBCZAK J P, et al. Membrane sculpting by curved DNA origami scaffolds [J]. Nature
Communications, 2018, 9: 811.
[51] GROME M W, ZHANG Z, PINCET F, et al. Vesicle tubulation with self-assembling DNA nanosprings [J]. Angewandte Chemie-
International Edition, 2018, 57: 5330-5334.
[52] GROME M W, ZHANG Z, LIN C. Stiffness and membrane anchor density modulate DNA-nanospring-induced vesicle tubulation [J]. ACS Appl Mater Interfaces, 2019, 11: 22987-22992.
[53] JOURNOT C M A, RAMAKRISHNA V, WALLACE M I, et al. Modifying membrane morphology and interactions with DNA
origami clathrin-mimic networks [J]. ACS Nano, 2019, 13: 9973-9979.
[54] YANG Y, WANG J, SHIGEMATSU H, et al. Self-assembly of sizecontrolled liposomes on DNA nanotemplates [J]. Nature Chemistry, 2016, 8: 476-483.
[55] ZHANG Z, YANG Y, PINCET F, et al. Placing and shaping liposomes with reconfigurable DNA nanocages [J]. Nature
Chemistry, 2017, 9: 653-659.
[56] PERRAULT S D, SHIH W M. Virus-inspired membrane encapsulation of DNA nanostructures to achieve in vivo stability [J].
ACS Nano, 2014, 8: 5132-5140.
[57] ZHAO Z, ZHANG M, HOGLE J M, et al. DNA-corralled nanodiscs for the structural and functional characterization of membrane
proteins and viral entry [J]. Journal of the American Chemical Society, 2018, 140: 10639-10643.
[58] XU W, NATHWANI B, LIN C, et al. A programmable DNA origami platform to organize SNAREs for membrane fusion [J]. Journal of the American Chemical Society, 2016, 138: 4439-4447.
[59] BIAN X, ZHANG Z, XIONG Q, et al. A programmable DNAorigami platform for studying lipid transfer between bilayers [J].
Nat Chem Biol, 2019, 15: 830-837.
[60] YANG Y, WU Z, WANG L, et al. Sorting sub-150-nm liposomes of distinct sizes by DNA-brick-assisted centrifugation [J]. Nat Chem, 2021, 13: 335-342.
[61] LANGECKER M, ARNAUT V, MARTIN T G, et al. Synthetic lipid membrane channels formed by designed DNA nanostructures [J]. Science, 2012, 338: 932-936.
[62] BURNS J R, GÖPFRICH K, WOOD J W, et al. Lipid-bilayerspanning DNA nanopores with a bifunctional porphyrin anchor [J].
Angewandte Chemie-International Edition, 2013, 52: 12069-12072.
[63] GÖPFRICH K, ZETTL T, MEIJERING A E C, et al. DNA-tile structures induce ionic currents through lipid membranes [J]. Nano
Letters, 2015, 15: 3134-3138.
[64] GÖPFRICH K, LI C-Y, RICCI M, et al. Large-conductance transmembrane porin made from DNA origami [J]. ACS Nano,
2016, 10: 8207-8214.
[65] KRISHNAN S, ZIEGLER D, ARNAUT V, et al. Molecular transport through large-diameter DNA nanopores [J]. Nature
Communications, 2016, 7: 12787.
[66] BURNS J R, SEIFERT A, FERTIG N, et al. A biomimetic DNAbased channel for the ligand-controlled transport of charged
molecular cargo across a biological membrane [J]. Nature Nanotechnology, 2016, 11: 152-156.
[67] LANPHERE C, OFFENBARTL-STIEGERT D, DOREY A, et al. Design, assembly, and characterization of membrane-spanning DNA
nanopores [J]. Nat Protoc, 2021, 16: 86-130.
[68] XING Y, DOREY A, JAYASINGHE L, et al. Highly shape- and size-tunable membrane nanopores made with DNA [J]. Nature
Nanotechnology, 2022, 17: 708-713.
/
| 〈 |
|
〉 |